|
The Crookes tubes require a small amount of air in them to function, from about 10 −6 to 5×10 −8 atmosphere (7×10 −4 - 4×10 −5 torr or 0.1-0.006 pascal). Instead, electrons are generated by the ionization of the residual air by a high DC voltage (from a few kilovolts to about 100 kilovolts) applied between the cathode and anode electrodes in the tube, usually by an induction coil (a "Ruhmkorff coil"). 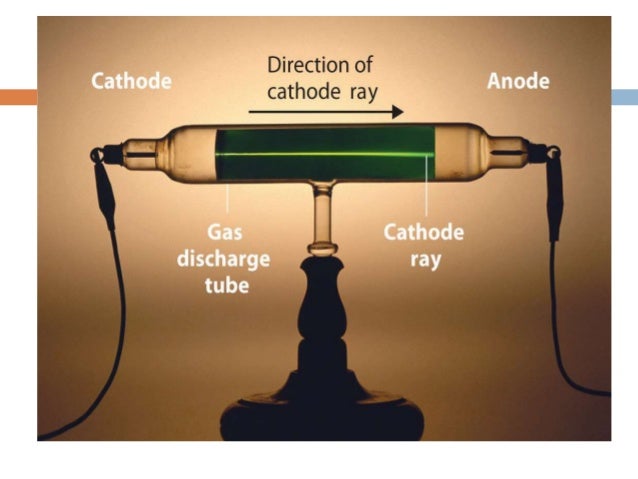
The term Crookes tube is also used for the first generation, cold cathode X-ray tubes, which evolved from the experimental Crookes tubes and were used until about 1920.Ĭrookes tubes are cold cathode tubes, meaning that they do not have a heated filament in them that releases electrons as the later electronic vacuum tubes usually do. Wilhelm Röntgen discovered X-rays using the Crookes tube in 1895. Crookes tubes are now used only for demonstrating cathode rays. Thomson's 1897 identification of cathode rays as negatively charged particles, which were later named electrons. 
It was used by Crookes, Johann Hittorf, Julius Plücker, Eugen Goldstein, Heinrich Hertz, Philipp Lenard, Kristian Birkeland and others to discover the properties of cathode rays, culminating in J.J. When a high voltage is applied between the electrodes, cathode rays ( electrons) are projected in straight lines from the cathode. ĭeveloped from the earlier Geissler tube, the Crookes tube consists of a partially evacuated glass bulb of various shapes, with two metal electrodes, the cathode and the anode, one at either end. The anode is the electrode at the bottom.Ī Crookes tube (also Crookes–Hittorf tube) is an early experimental electrical discharge tube, with partial vacuum, invented by English physicist William Crookes and others around 1869-1875, in which cathode rays, streams of electrons, were discovered. Electrons (cathode rays) travel in straight lines from the cathode (left), as shown by the shadow cast by the metal Maltese cross on the fluorescence of the righthand glass wall of the tube. Electrons are the basic units of all atoms.A Crookes tube: light and dark. The behaviour of cathode rays is similar to negatively charged particles (electrons) in the presence of an electrical or a magnetic field.Ĭharacteristics of cathode rays do not depend upon: the material of the electrodes and the nature of the gas present in the tube These rays travel in a straight line in the absence of electric and magnetic fields. They can be observed with the help of phosphorescent or fluorescent materials (such as zinc sulphide). Stream of particles move from the cathode (− ve) to the anode (+ ve) −−− known as cathode rays or cathode ray particlesĬathode rays move from the cathode to the anode.Ĭathode rays are not visible. 
The glass tube is partially evacuated (Low pressure inside the tube) Very high voltage is applied across the electrodes The brief explanation for the discovery of electron is described as:ĭiscovery of Electron (Michael Faraday’s Cathode Ray Discharge Tube Experiment) They performed experiments indicating that cathode rays really were unique particles, rather than waves, atoms, or molecules as was believed earlier. Thomson and his team of British physicists. 1) The electron was identified and assigned the negative charge in 1897 by J.J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
